University of California, San Diego
Physics 1b - Thermal Physics & Electromagnetism

 |
Physics 1B - Tutorial #2
|

- Very cold ice is placed in a closed container. Heat is then applied to the
container. Below is a graph of the temperature inside the container
plotted against the amount of heat delivered to the system.
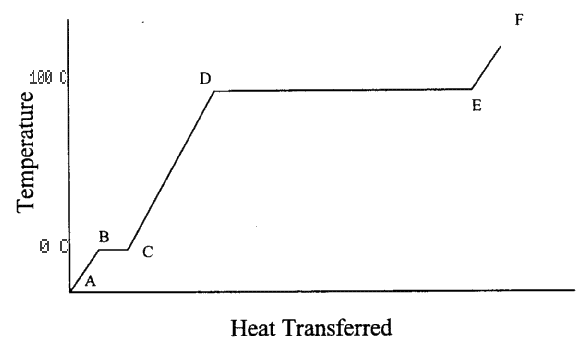
- Describe what is happening to the substance inside the container from
point A to point F.
- A --> B ice temperature rising as heat enters container.
- B --> C ice is melting; heat --> heat of fusion necessary to melt ice.
reached.
- C --> D water temperature is rising.
- D --> E water is evaporating; heat --> heat of vaporization.
- E --> F water vapor temperature is rising.
Put temperature scales in Celsius on the vertical axis.
- B --> C = 0° C
- D --> E = 100° C
as above.
If the heating rate is constant (dQ/dt = constant), compare the time it
takes to heat from B to C to the time it takes to heat from D to E.
The latent heat of water vaporizing (heat of vaporization;
Lv = 540 kcal/kg) is 6.75 times the latent heat of ice melting
(heat of fusion; Lf = 80 kcal/kg), D --> E takes 6.75 times
as long as the time from B --> C.
Materials A and B have equal densities. But A has twice the specific heat
of B. You have two 100 g cubes made of materials A and B.
- Cube A, initially at 0°C, is placed in good thermal contact
with cube B, initially at 100°C. Both are placed in a
well-insulated container. What is the final temperature?
CA(100g)(T-0)=CB(100g)(100-T)
CA=2CB
2x(100g)(T-0)=(100g)(100-T)
Solving: T=33.3 °C.
Cubes A and B are both heated to 200°C, and then placed in
room temperature environment. Which one cools faster?
The two cubes have the same surface area. Their heat loss rates are the same.
Cube B has a lower heat capacity. Cube B cools faster.
A thermometer is laid out in direct sunlight. Does it measure the
temperature of the air, of the sun, or of something else?
The thermometer always measures the temperature of the thermometer.
Its temperature is indicative of the temperature of another object only if
it is in thermal equilibrium with the object (that's why your Mother makes
you hold the thermometer under your tongue for so long - to establish
thermal equilibrium. In principle, if the thermometer had perfect
thermal radiative properties (called a blackbody) it would reach
thermal equilbrium with the sun by radiative processes. Since the thermometer
doesn't have perfect radiative properties (for one thing it would melt before
reaching 6000K), in this case, its temperature is neither that of the sun nor
that of the air.
The inside of an oven is at 400° F. But you can still put
your hand in the oven as long as you don't touch anything. But since the
air inside the oven is also at 400°F, why isn't your hand
burned just the same? What if you leave your hand in the oven for a
long time (e.g. 2 hours)?
Air is a very poor thermal conductor. (thousands of times less than that
of iron). Therefore, its heat cannot be effectively transferred to your hand.
If you touch the shelves made of iron, heat can be transferred quickly and you
will be burned. If you leave your hand in the oven for too long, however, even
just the air will transfer enough thermal energy to roast you.
While jogging, an average 65 kg student generates thermal energy at a
rate of 300 W (about 0.4 horse power). To maintain a constant body temperature
of 37°C, this energy has to be dissipated by perspiration or
other mechanisms.
- If these mechanisms failed and the heat couldn't flow out of
the student's body, for how long could a student run before irreversible body
damage could occur? (Protein structures in the body are irreversibly damaged
at 44°C or above. The specific heat capacity of a typical human
body is 3480 J/kg.K, slightly less than that of water.)
At a heating rate of 300W, it takes
(3480J/kg/K)(65kg)(7K)/(300W)=5280s=1.5 hour
to raise the body temperature of the student by 7K, or 7 °C.
If the heat loss is by sweat evaporation only, calculate the volume of
water evaporated per minute (latent heat Lv = 539 cal/g). How many
liters of water are needed to
compensate the water loss of a two hour jog?
Latent heat Lv=539 cal/g=2.26 kJ/g. To dissipate at a rate of 300W, water has
to evaporate at
(300W)/(2.26 kJ/g)=0.13g/s, or 0.13 ml per second, or 8 ml/min.
A 2-hour jog requires an intake of (8ml/min)(120min)=1 liter
water.
What other heat loss mechanisms do you think may be important for a
runner?
The body looses heat due to breathing. Cold air is inhaled and warm air
exhaled. The exhaled air is also more moist than the inhaled air. This
water loss is actually part of the evaporated water calculated above. Heat
is also lost due to conduction (from warm skin to cold air), and to
convection as that warmed air rises and is replaced by cold air. Since the
body is (usually) hotter than its surroundings more heat is lost due to
radiation emitted to the surroundings than is gained from radiation
absorbed from the surroundings.
Within the body heat is transferred mainly by blood flow. Blood is warmed
as it passes through the heart and lungs, and cooled at the body surface.
Blood capillaries near the skin open when the body wishes to get rid of heat
(eg jogging or sitting in the sauna), and close when we get cold and want
to conserve body heat.

 Physics 1B Home
Physics 1B Home
 Tutorials
Tutorials
 Tutorial #1
Tutorial #1
 Tutorial #3
Tutorial #3
Gene Smith
Last
modified: Thurs., 13 Apr 2000




![]() Physics 1B Home
Physics 1B Home
![]() Tutorials
Tutorials
![]() Tutorial #1
Tutorial #1
![]() Tutorial #3
Tutorial #3